| |
“Ozone chemistry is at the heart of atmospheric chemistry.” Bill Stockwell, Desert Research Institute
Ozone affects climate, and climate affects ozone. Temperature, humidity, winds, and the presence of other chemicals in the atmosphere influence ozone formation, and the presence of ozone, in turn, affects those atmospheric constituents.
Interactions between ozone and climate have been subjects of discussion ever since the early 1970s when scientists first suggested that human-produced chemicals could destroy our ozone shield in the upper atmosphere. The discussion intensified in 1985 when atmospheric scientists discovered an ozone “hole” in the upper atmosphere (stratosphere) over Antarctica. Today, some scientists are predicting the stratospheric ozone layer will recover to 1980 ozone levels by the year 2050. These scientists say we can expect recovery by that time because most nations have been abiding by international agreements to phase out production of ozone-depleting chemicals such as chlorofluorocarbons (CFCs) and halons. But the atmosphere continues to surprise us, and some atmospheric scientists recently demonstrated a new spin on the ozone recovery story that may change its ending. Well before the expected stratospheric ozone layer recovery date of 2050, ozone’s effects on climate may become the main driver of ozone loss in the stratosphere. As a result, ozone recovery may not be complete until 2060 or 2070. |
|
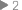 |
| |

“I was looking the other way when it hit me,” says Drew Shindell, physicist at the NASA Goddard Institute for Space Studies (GISS). “I was aware that the stratosphere was cooling, and I was wondering about the effects that cooling was having on the chemistry of the atmosphere, especially on ozone depletion.” That is, initially he was wondering about the effects that climate might be having on ozone, not the effects that ozone itself might be having on climate.
Ozone’s impact on climate consists primarily of changes in temperature. The more ozone in a given parcel of air, the more heat it retains. Ozone generates heat in the stratosphere, both by absorbing the sun’s ultraviolet radiation and by absorbing upwelling infrared radiation from the lower atmosphere (troposphere). Consequently, decreased ozone in the stratosphere results in lower temperatures. Observations show that over recent decades, the mid to upper stratosphere (from 30 to 50 km above the Earth’s surface) has cooled by 1° to 6° C (2° to 11° F). This stratospheric cooling has taken place at the same time that greenhouse gas amounts in the lower atmosphere (troposphere) have risen. The two phenomena may be linked.
Shindell says, “I’ve long been aware that chemistry and climate influence one another strongly. I started to ask how cold the stratosphere might get because of increasing amounts of greenhouse gases. I was wondering whether or not the cooling in the stratosphere would be rapid enough that more ozone depletion would take place than we had previously calculated. Would the cooling be so fast that even more ozone depletion would occur before the impact of international agreements to limit ozone had time to take effect?” This would create a possible feedback loop. The more ozone destruction in the stratosphere, the colder it would get just because there was less ozone. And the colder it would get, the more ozone depletion would occur. |
|
A midnight sun shines across the frigid Antarctic landscape, and makes dark triangles of researchers’ tents. Low temperatures in the stratosphere above both the Antarctic and the Arctic contribute to ozone depletion. Since ozone depletion itself contributes to decreasing temperatures, a feedback loop may be in operation. (Photograph courtesy Alberto Behar, NASA JPL/National Science Foundation) |
| |
The deepest ozone losses over both the Arctic and the Antarctic result from special conditions that occur in the winter and early spring. As winter arrives, a vortex of winds develops around the pole and isolates the polar stratosphere. When temperatures drop below -78°C (-109°F), thin clouds form of ice, nitric acid, and sulphuric acid mixtures. Chemical reactions on the surfaces of ice crystals in the clouds release active forms of CFCs. Ozone depletion begins, and the ozone “hole” appears. In spring, temperatures begin to rise, the ice evaporates, and the ozone layer starts to recover. |
|
|
| |

 Reflections on a Possible Delay Reflections on a Possible Delay
|
|
Thin clouds made of mixtures of ice, nitric acid, and sulphuric acid form in the upper atmosphere (stratosphere) over the poles when temperatures drop below -78°C (-109°F). Ozone depletion occurs in such polar stratospheric clouds. This particular cloud formation appeared over Iceland at an altitude of about 22 km on February 4, 2003. Its beautiful colors result from the very small size of the ice crystals it contains. These tiny particles bend and scatter the sun’s rays much like a prism. (Photograph courtesy Mark R. Schoeberl, NASA GSFC) |

