

The Earth’s climate is a solar powered system. Globally, over the course of the year, the Earth system—land surfaces, oceans, and atmosphere—absorbs an average of about 240 watts of solar power per square meter (one watt is one joule of energy every second). The absorbed sunlight drives photosynthesis, fuels evaporation, melts snow and ice, and warms the Earth system.

Solar power drives Earth’s climate. Energy from the Sun heats the surface, warms the atmosphere, and powers the ocean currents. (Astronaut photograph ISS015-E-10469, courtesy NASA/JSC Gateway to Astronaut Photography of Earth.)
The Sun doesn’t heat the Earth evenly. Because the Earth is a sphere, the Sun heats equatorial regions more than polar regions. The atmosphere and ocean work non-stop to even out solar heating imbalances through evaporation of surface water, convection, rainfall, winds, and ocean circulation. This coupled atmosphere and ocean circulation is known as Earth’s heat engine.
The climate’s heat engine must not only redistribute solar heat from the equator toward the poles, but also from the Earth’s surface and lower atmosphere back to space. Otherwise, Earth would endlessly heat up. Earth’s temperature doesn’t infinitely rise because the surface and the atmosphere are simultaneously radiating heat to space. This net flow of energy into and out of the Earth system is Earth’s energy budget.
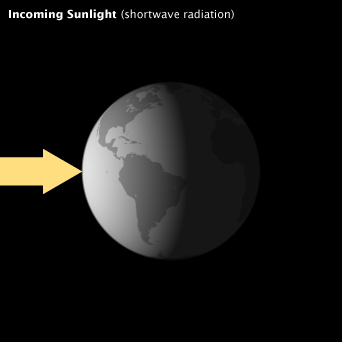
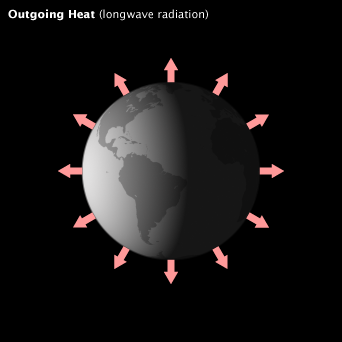
When the flow of incoming solar energy is balanced by an equal flow of heat to space, Earth is in radiative equilibrium, and global temperature is relatively stable. Anything that increases or decreases the amount of incoming or outgoing energy disturbs Earth’s radiative equilibrium; global temperatures rise or fall in response.
All matter in the universe that has a temperature above absolute zero (the temperature at which all atomic or molecular motion stops) radiates energy across a range of wavelengths in the electromagnetic spectrum. The hotter something is, the shorter its peak wavelength of radiated energy is. The hottest objects in the universe radiate mostly gamma rays and x-rays. Cooler objects emit mostly longer-wavelength radiation, including visible light, thermal infrared, radio, and microwaves.
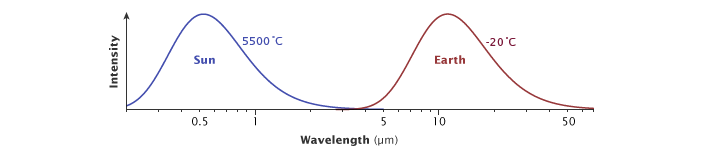
The Sun’s surface temperature is 5,500° C, and its peak radiation is in visible wavelengths of light. Earth’s effective temperature—the temperature it appears when viewed from space—is -20° C, and it radiates energy that peaks in thermal infrared wavelengths. (Illustration adapted from Robert Rohde.)

Incandescent light bulbs radiate 40 to 100 watts. The Sun delivers 1,360 watts per square meter. An astronaut facing the Sun has a surface area of about 0.85 square meters, so he or she receives energy equivalent to 19 60-watt light bulbs. (Photograph ©2005 Paul Watson.)
The surface of the Sun has a temperature of about 5,800 Kelvin (about 5,500 degrees Celsius, or about 10,000 degrees Fahrenheit). At that temperature, most of the energy the Sun radiates is visible and near-infrared light. At Earth’s average distance from the Sun (about 150 million kilometers), the average intensity of solar energy reaching the top of the atmosphere directly facing the Sun is about 1,360 watts per square meter, according to measurements made by the most recent NASA satellite missions. This amount of power is known as the total solar irradiance. (Before scientists discovered that it varies by a small amount during the sunspot cycle, total solar irradiance was sometimes called “the solar constant.”)
A watt is measurement of power, or the amount of energy that something generates or uses over time. How much power is 1,360 watts? An incandescent light bulb uses anywhere from 40 to 100 watts. A microwave uses about 1000 watts. If for just one hour, you could capture and re-use all the solar energy arriving over a single square meter at the top of the atmosphere directly facing the Sun—an area no wider than an adult’s outstretched arm span—you would have enough to run a refrigerator all day.
The total solar irradiance is the maximum possible power that the Sun can deliver to a planet at Earth’s average distance from the Sun; basic geometry limits the actual solar energy intercepted by Earth. Only half the Earth is ever lit by the Sun at one time, which halves the total solar irradiance.
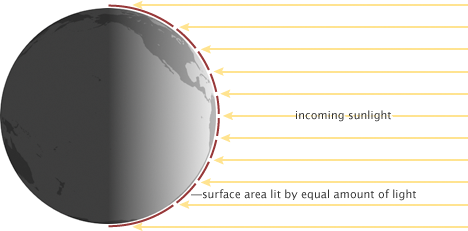
Energy from sunlight is not spread evenly over Earth. One hemisphere is always dark, receiving no solar radiation at all. On the daylight side, only the point directly under the Sun receives full-intensity solar radiation. From the equator to the poles, the Sun’ rays meet Earth at smaller and smaller angles, and the light gets spread over larger and larger surface areas (red lines). (NASA illustration by Robert Simmon.)
In addition, the total solar irradiance is the maximum power the Sun can deliver to a surface that is perpendicular to the path of incoming light. Because the Earth is a sphere, only areas near the equator at midday come close to being perpendicular to the path of incoming light. Everywhere else, the light comes in at an angle. The progressive decrease in the angle of solar illumination with increasing latitude reduces the average solar irradiance by an additional one-half.
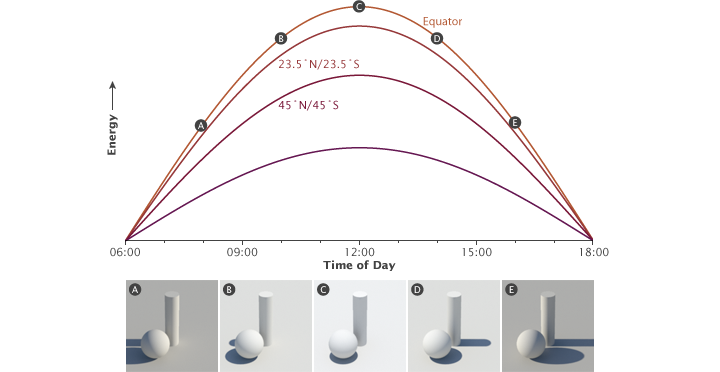
The solar radiation received at Earth’s surface varies by time and latitude. This graph illustrates the relationship between latitude, time, and solar energy during the equinoxes. The illustrations show how the time of day (A-E) affects the angle of incoming sunlight (revealed by the length of the shadow) and the light’s intensity. On the equinoxes, the Sun rises at 6:00 a.m. everywhere. The strength of sunlight increases from sunrise until noon, when the Sun is directly overhead along the equator (casting no shadow). After noon, the strength of sunlight decreases until the Sun sets at 6:00 p.m. The tropics (from 0 to 23.5° latitude) receive about 90% of the energy compared to the equator, the mid-latitudes (45°) roughly 70%, and the Arctic and Antarctic Circles about 40%. (NASA illustration by Robert Simmon.)
Averaged over the entire planet, the amount of sunlight arriving at the top of Earth’s atmosphere is only one-fourth of the total solar irradiance, or approximately 340 watts per square meter.
When the flow of incoming solar energy is balanced by an equal flow of heat to space, Earth is in radiative equilibrium, and global temperature is relatively stable. Anything that increases or decreases the amount of incoming or outgoing energy disturbs Earth’s radiative equilibrium; global temperatures must rise or fall in response.
Three hundred forty watts per square meter of incoming solar power is a global average; solar illumination varies in space and time. The annual amount of incoming solar energy varies considerably from tropical latitudes to polar latitudes (described on page 2). At middle and high latitudes, it also varies considerably from season to season.
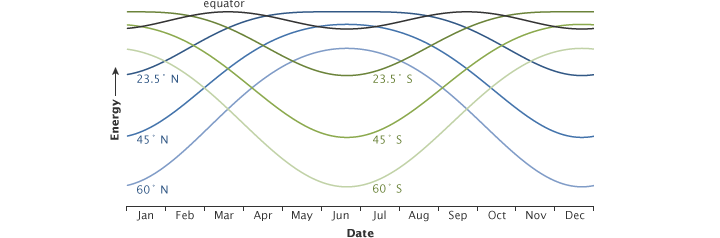
The peak energy received at different latitudes changes throughout the year. This graph shows how the solar energy received at local noon each day of the year changes with latitude. At the equator (gray line), the peak energy changes very little throughout the year. At high northern (blue lines) and southern (green) latitudes, the seasonal change is extreme. (NASA illustration by Robert Simmon.)
If the Earth’s axis of rotation were vertical with respect to the path of its orbit around the Sun, the size of the heating imbalance between equator and the poles would be the same year round, and the seasons we experience would not occur. Instead Earth’s axis is tilted off vertical by about 23 degrees. As the Earth orbits the Sun, the tilt causes one hemisphere and then the other to receive more direct sunlight and to have longer days.
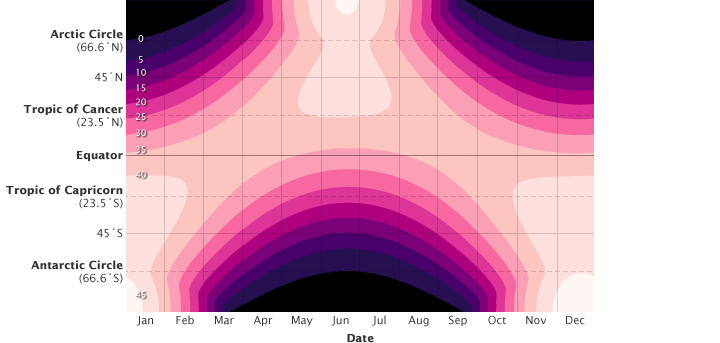
The total energy received each day at the top of the atmosphere depends on latitude. The highest daily amounts of incoming energy (pale pink) occur at high latitudes in summer, when days are long, rather than at the equator. In winter, some polar latitudes receive no light at all (black). The Southern Hemisphere receives more energy during December (southern summer) than the Northern Hemisphere does in June (northern summer) because Earth’s orbit is not a perfect circle and Earth is slightly closer to the Sun during that part of its orbit. Total energy received ranges from 0 (during polar winter) to about 50 (during polar summer) megajoules per square meter per day.
In the “summer hemisphere,” the combination of more direct sunlight and longer days means the pole can receive more incoming sunlight than the tropics, but in the winter hemisphere, it gets none. Even though illumination increases at the poles in the summer, bright white snow and sea ice reflect a significant portion of the incoming light, reducing the potential solar heating.
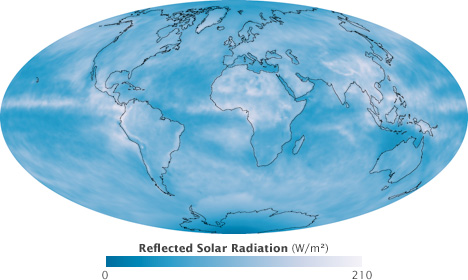
The amount of sunlight the Earth absorbs depends on the reflectivness of the atmosphere and the ground surface. This satellite map shows the amount of solar radiation (watts per square meter) reflected during September 2008. Along the equator, clouds reflected a large proportion of sunlight, while the pale sands of the Sahara caused the high reflectivness in North Africa. Neither pole is receiving much incoming sunlight at this time of year, so they reflect little energy even though both are ice-covered. (NASA map by Robert Simmon, based on CERES data.)
The differences in reflectivness (albedo) and solar illumination at different latitudes lead to net heating imbalances throughout the Earth system. At any place on Earth, the net heating is the difference between the amount of incoming sunlight and the amount heat radiated by the Earth back to space (for more on this energy exchange see Page 4). In the tropics there is a net energy surplus because the amount of sunlight absorbed is larger than the amount of heat radiated. In the polar regions, however, there is an annual energy deficit because the amount of heat radiated to space is larger than the amount of absorbed sunlight.
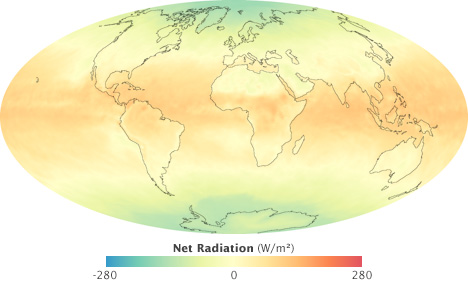
This map of net radiation (incoming sunlight minus reflected light and outgoing heat) shows global energy imbalances in September 2008, the month of an equinox. Areas around the equator absorbed about 200 watts per square meter more on average (orange and red) than they reflected or radiated. Areas near the poles reflected and/or radiated about 200 more watts per square meter (green and blue) than they absorbed. Mid-latitudes were roughly in balance. (NASA map by Robert Simmon, based on CERES data.)
The net heating imbalance between the equator and poles drives an atmospheric and oceanic circulation that climate scientists describe as a “heat engine.” (In our everyday experience, we associate the word engine with automobiles, but to a scientist, an engine is any device or system that converts energy into motion.) The climate is an engine that uses heat energy to keep the atmosphere and ocean moving. Evaporation, convection, rainfall, winds, and ocean currents are all part of the Earth’s heat engine.
Note: Determining exact values for energy flows in the Earth system is an area of ongoing climate research. Different estimates exist, and all estimates have some uncertainty. Estimates come from satellite observations, ground-based observations, and numerical weather models. The numbers in this article rely most heavily on direct satellite observations of reflected sunlight and thermal infrared energy radiated by the atmosphere and the surface.
Earth’s heat engine does more than simply move heat from one part of the surface to another; it also moves heat from the Earth’s surface and lower atmosphere back to space. This flow of incoming and outgoing energy is Earth’s energy budget. For Earth’s temperature to be stable over long periods of time, incoming energy and outgoing energy have to be equal. In other words, the energy budget at the top of the atmosphere must balance. This state of balance is called radiative equilibrium.
About 29 percent of the solar energy that arrives at the top of the atmosphere is reflected back to space by clouds, atmospheric particles, or bright ground surfaces like sea ice and snow. This energy plays no role in Earth’s climate system. About 23 percent of incoming solar energy is absorbed in the atmosphere by water vapor, dust, and ozone, and 48 percent passes through the atmosphere and is absorbed by the surface. Thus, about 71 percent of the total incoming solar energy is absorbed by the Earth system.
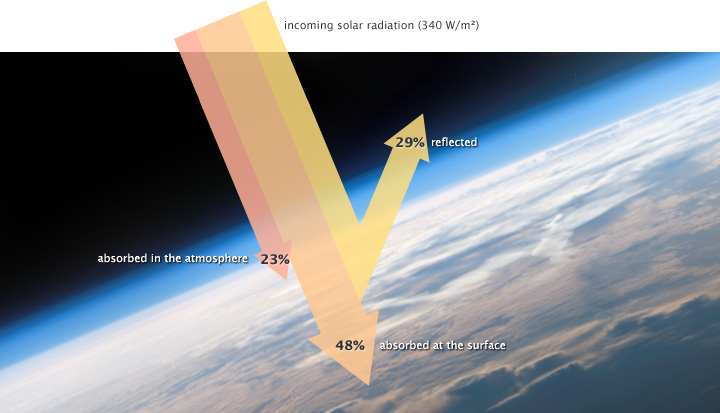
Of the 340 watts per square meter of solar energy that falls on the Earth, 29% is reflected back into space, primarily by clouds, but also by other bright surfaces and the atmosphere itself. About 23% of incoming energy is absorbed in the atmosphere by atmospheric gases, dust, and other particles. The remaining 48% is absorbed at the surface. (NASA illustration by Robert Simmon. Astronaut photograph ISS013-E-8948.)
When matter absorbs energy, the atoms and molecules that make up the material become excited; they move around more quickly. The increased movement raises the material’s temperature. If matter could only absorb energy, then the temperature of the Earth would be like the water level in a sink with no drain where the faucet runs continuously.
Temperature doesn’t infinitely rise, however, because atoms and molecules on Earth are not just absorbing sunlight, they are also radiating thermal infrared energy (heat). The amount of heat a surface radiates is proportional to the fourth power of its temperature. If temperature doubles, radiated energy increases by a factor of 16 (2 to the 4th power). If the temperature of the Earth rises, the planet rapidly emits an increasing amount of heat to space. This large increase in heat loss in response to a relatively smaller increase in temperature—referred to as radiative cooling—is the primary mechanism that prevents runaway heating on Earth.
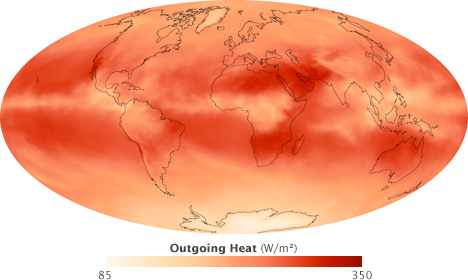
Absorbed sunlight is balanced by heat radiated from Earth’s surface and atmosphere. This satellite map shows the distribution of thermal infrared radiation emitted by Earth in September 2008. Most heat escaped from areas just north and south of the equator, where the surface was warm, but there were few clouds. Along the equator, persistent clouds prevented heat from escaping. Likewise, the cold poles radiated little heat. (NASA map by Robert Simmon, based on CERES data.)
The atmosphere and the surface of the Earth together absorb 71 percent of incoming solar radiation, so together, they must radiate that much energy back to space for the planet’s average temperature to remain stable. However, the relative contribution of the atmosphere and the surface to each process (absorbing sunlight versus radiating heat) is asymmetric. The atmosphere absorbs 23 percent of incoming sunlight while the surface absorbs 48. The atmosphere radiates heat equivalent to 59 percent of incoming sunlight; the surface radiates only 12 percent. In other words, most solar heating happens at the surface, while most radiative cooling happens in the atmosphere. How does this reshuffling of energy between the surface and atmosphere happen?
To understand how the Earth’s climate system balances the energy budget, we have to consider processes occurring at the three levels: the surface of the Earth, where most solar heating takes place; the edge of Earth’s atmosphere, where sunlight enters the system; and the atmosphere in between. At each level, the amount of incoming and outgoing energy, or net flux, must be equal.
Remember that about 29 percent of incoming sunlight is reflected back to space by bright particles in the atmosphere or bright ground surfaces, which leaves about 71 percent to be absorbed by the atmosphere (23 percent) and the land (48 percent). For the energy budget at Earth’s surface to balance, processes on the ground must get rid of the 48 percent of incoming solar energy that the ocean and land surfaces absorb. Energy leaves the surface through three processes: evaporation, convection, and emission of thermal infrared energy.
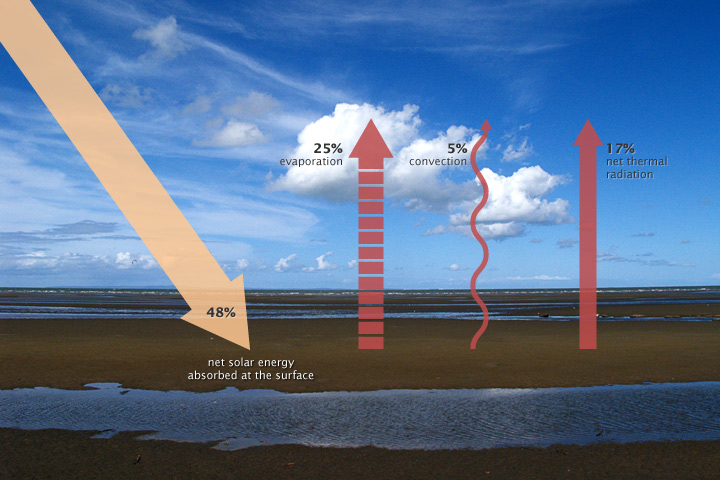
The surface absorbs about 48% of incoming sunlight. Three processes remove an equivalent amount of energy from the Earth’s surface: evaporation (25%), convection (5%), and thermal infrared radiation, or heat (net 17%). (NASA illustration by Robert Simmon. Photograph ©2006 Cyron.)
About 25 percent of incoming solar energy leaves the surface through evaporation. Liquid water molecules absorb incoming solar energy, and they change phase from liquid to gas. The heat energy that it took to evaporate the water is latent in the random motions of the water vapor molecules as they spread through the atmosphere. When the water vapor molecules condense back into rain, the latent heat is released to the surrounding atmosphere. Evaporation from tropical oceans and the subsequent release of latent heat are the primary drivers of the atmospheric heat engine (described on page 3).

Towers of cumulus clouds transport energy away from the surface of the Earth. Solar heating drives evaporation. Warm, moist air becomes buoyant and rises, moving energy from the surface high into the atmosphere. Energy is released back into the atmosphere when the water vapor condenses into liquid water or freezes into ice crystals. (Astronaut Photograph ISS006-E-19436.)
An additional 5 percent of incoming solar energy leaves the surface through convection. Air in direct contact with the sun-warmed ground becomes warm and buoyant. In general, the atmosphere is warmer near the surface and colder at higher altitudes, and under these conditions, warm air rises, shuttling heat away from the surface.
Finally, a net of about 17 percent of incoming solar energy leaves the surface as thermal infrared energy (heat) radiated by atoms and molecules on the surface. This net upward flux results from two large but opposing fluxes: heat flowing upward from the surface to the atmosphere (117%) and heat flowing downward from the atmosphere to the ground (100%). (These competing fluxes are part of the greenhouse effect, described on page 6.) Remember that the peak wavelength of energy a surface radiates is based on its temperature. The Sun’s peak radiation is at visible and near-infrared wavelengths. The Earth’s surface is much cooler, only about 15 degrees Celsius on average. The peak radiation from the surface is at thermal infrared wavelengths around 12.5 micrometers.
Just as the incoming and outgoing energy at the Earth’s surface must balance, the flow of energy into the atmosphere must be balanced by an equal flow of energy out of the atmosphere and back to space. Satellite measurements indicate that the atmosphere radiates thermal infrared energy equivalent to 59 percent of the incoming solar energy. If the atmosphere is radiating this much, it must be absorbing that much. Where does that energy come from?
Clouds, aerosols, water vapor, and ozone directly absorb 23 percent of incoming solar energy. Evaporation and convection transfer 25 and 5 percent of incoming solar energy from the surface to the atmosphere. These three processes transfer the equivalent of 53 percent of the incoming solar energy to the atmosphere. If total inflow of energy must match the outgoing thermal infrared observed at the top of the atmosphere, where does the remaining fraction (about 5-6 percent) come from? The remaining energy comes from the Earth’s surface.
Just as the major atmospheric gases (oxygen and nitrogen) are transparent to incoming sunlight, they are also transparent to outgoing thermal infrared. However, water vapor, carbon dioxide, methane, and other trace gases are opaque to many wavelengths of thermal infrared energy. Remember that the surface radiates the net equivalent of 17 percent of incoming solar energy as thermal infrared. However, the amount that directly escapes to space is only about 12 percent of incoming solar energy. The remaining fraction—a net 5-6 percent of incoming solar energy—is transferred to the atmosphere when greenhouse gas molecules absorb thermal infrared energy radiated by the surface.

The atmosphere radiates the equivalent of 59% of incoming sunlight back to space as thermal infrared energy, or heat. Where does the atmosphere get its energy? The atmosphere directly absorbs about 23% of incoming sunlight, and the remaining energy is transferred from the Earth’s surface by evaporation (25%), convection (5%), and thermal infrared radiation (a net of 5-6%). The remaining thermal infrared energy from the surface (12%) passes through the atmosphere and escapes to space. (NASA illustration by Robert Simmon. Astronaut photograph ISS017-E-13859.)
When greenhouse gas molecules absorb thermal infrared energy, their temperature rises. Like coals from a fire that are warm but not glowing, greenhouse gases then radiate an increased amount of thermal infrared energy in all directions. Heat radiated upward continues to encounter greenhouse gas molecules; those molecules absorb the heat, their temperature rises, and the amount of heat they radiate increases. At an altitude of roughly 5-6 kilometers, the concentration of greenhouse gases in the overlying atmosphere is so small that heat can radiate freely to space.
Because greenhouse gas molecules radiate heat in all directions, some of it spreads downward and ultimately comes back into contact with the Earth’s surface, where it is absorbed. The temperature of the surface becomes warmer than it would be if it were heated only by direct solar heating. This supplemental heating of the Earth’s surface by the atmosphere is the natural greenhouse effect.
The natural greenhouse effect raises the Earth’s surface temperature to about 15 degrees Celsius on average—more than 30 degrees warmer than it would be if it didn’t have an atmosphere. The amount of heat radiated from the atmosphere to the surface (sometimes called “back radiation”) is equivalent to 100 percent of the incoming solar energy. The Earth’s surface responds to the “extra” (on top of direct solar heating) energy by raising its temperature.
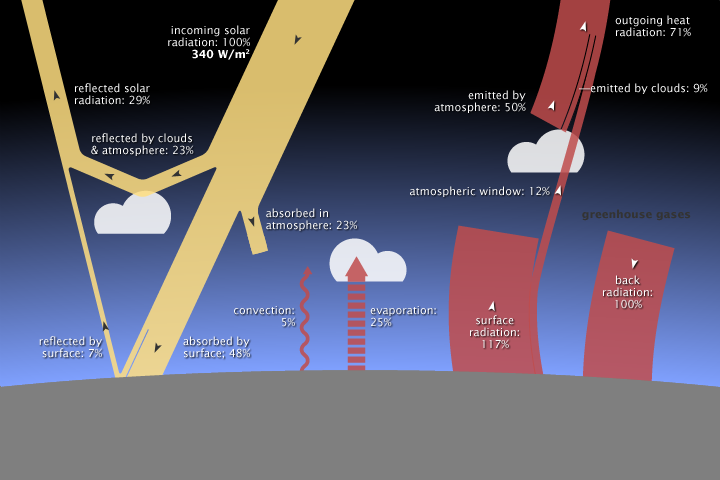
On average, 340 watts per square meter of solar energy arrives at the top of the atmosphere. Earth returns an equal amount of energy back to space by reflecting some incoming light and by radiating heat (thermal infrared energy). Most solar energy is absorbed at the surface, while most heat is radiated back to space by the atmosphere. Earth's average surface temperature is maintained by two large, opposing energy fluxes between the atmosphere and the ground (right)—the greenhouse effect. NASA illustration by Robert Simmon, adapted from Trenberth et al. 2009, using CERES flux estimates provided by Norman Loeb.)
Why doesn’t the natural greenhouse effect cause a runaway increase in surface temperature? Remember that the amount of energy a surface radiates always increases faster than its temperature rises—outgoing energy increases with the fourth power of temperature. As solar heating and “back radiation” from the atmosphere raise the surface temperature, the surface simultaneously releases an increasing amount of heat—equivalent to about 117 percent of incoming solar energy. The net upward heat flow, then, is equivalent to 17 percent of incoming sunlight (117 percent up minus 100 percent down).
Some of the heat escapes directly to space, and the rest is transferred to higher and higher levels of the atmosphere, until the energy leaving the top of the atmosphere matches the amount of incoming solar energy. Because the maximum possible amount of incoming sunlight is fixed by the solar constant (which depends only on Earth’s distance from the Sun and very small variations during the solar cycle), the natural greenhouse effect does not cause a runaway increase in surface temperature on Earth.
Any changes to the Earth’s climate system that affect how much energy enters or leaves the system alters Earth’s radiative equilibrium and can force temperatures to rise or fall. These destabilizing influences are called climate forcings. Natural climate forcings include changes in the Sun’s brightness, Milankovitch cycles (small variations in the shape of Earth’s orbit and its axis of rotation that occur over thousands of years), and large volcanic eruptions that inject light-reflecting particles as high as the stratosphere. Manmade forcings include particle pollution (aerosols), which absorb and reflect incoming sunlight; deforestation, which changes how the surface reflects and absorbs sunlight; and the rising concentration of atmospheric carbon dioxide and other greenhouse gases, which decrease heat radiated to space. A forcing can trigger feedbacks that intensify or weaken the original forcing. The loss of ice at the poles, which makes them less reflective, is an example of a feedback.



Carbon dioxide forces the Earth’s energy budget out of balance by absorbing thermal infrared energy (heat) radiated by the surface. It absorbs thermal infrared energy with wavelengths in a part of the energy spectrum that other gases, such as water vapor, do not. Although water vapor is a powerful absorber of many wavelengths of thermal infrared energy, it is almost transparent to others. The transparency at those wavelengths is like a window the atmosphere leaves open for radiative cooling of the Earth’s surface. The most important of these “water vapor windows” is for thermal infrared with wavelengths centered around 10 micrometers. (The maximum transparency occurs at 10 micrometers, but partial transparency occurs for wavelengths between about 8 and about 14 micrometers.)
Carbon dioxide is a very strong absorber of thermal infrared energy with wavelengths longer than 12-13 micrometers, which means that increasing concentrations of carbon dioxide partially “close” the atmospheric window. In other words, wavelengths of outgoing thermal infrared energy that our atmosphere’s most abundant greenhouse gas—water vapor—would have let escape to space are instead absorbed by carbon dioxide.
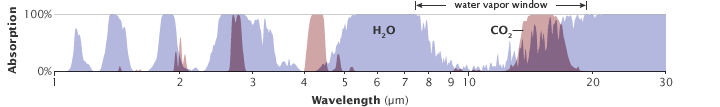
All atmospheric gases have a unique pattern of energy absorption: they absorb some wavelengths of energy but are transparent to others. The absorption patterns of water vapor (blue peaks) and carbon dioxide (pink peaks) overlap in some wavelengths. Carbon dioxide is not as strong a greenhouse gas as water vapor, but it absorbs energy in wavelengths (12-15 micrometers) that water vapor does not, partially closing the “window” through which heat radiated by the surface would normally escape to space. (Illustration adapted from Robert Rohde.)
The absorption of outgoing thermal infrared by carbon dioxide means that Earth still absorbs about 70 percent of the incoming solar energy, but an equivalent amount of heat is no longer leaving. The exact amount of the energy imbalance is very hard to measure, but it appears to be a little over 0.8 watts per square meter. The imbalance is inferred from a combination of measurements, including satellite and ocean-based observations of sea level rise and warming.
When a forcing like increasing greenhouse gas concentrations bumps the energy budget out of balance, it doesn’t change the global average surface temperature instantaneously. It may take years or even decades for the full impact of a forcing to be felt. This lag between when an imbalance occurs and when the impact on surface temperature becomes fully apparent is mostly because of the immense heat capacity of the global ocean. The heat capacity of the oceans gives the climate a thermal inertia that can make surface warming or cooling more gradual, but it can’t stop a change from occurring.
The changes we have seen in the climate so far are only part of the full response we can expect from the current energy imbalance, caused only by the greenhouse gases we have released so far. Global average surface temperature has risen between 0.6 and 0.9 degrees Celsius in the past century, and it will likely rise at least 0.6 degrees in response to the existing energy imbalance.
As the surface temperature rises, the amount of heat the surface radiates will increase rapidly (see description of radiative cooling on Page 4). If the concentration of greenhouse gases stabilizes, then Earth’s climate will once again come into equilibrium, albeit with the “thermostat”—global average surface temperature—set at a higher temperature than it was before the Industrial Revolution.
However, as long as greenhouse gas concentrations continue to rise, the amount of absorbed solar energy will continue to exceed the amount of thermal infrared energy that can escape to space. The energy imbalance will continue to grow, and surface temperatures will continue to rise.