

The title of this post is borrowed from Phillip Pullman’s prequel to his Dark Materials trilogy. I loved these books as a child, and by happy coincidence ended up studying for my masters at the college on which Jordan College in the novels is based. The reference to journeys northward and parallels with searching for particles struck me as appropriate.

View from the DC-8 toward the NASA-Armstrong hangar in Palmdale, California (Credit: Christina Williamson).
Pre-flight in the Californian desert heat at 4am and a sunrise take off felt very far removed from out aim of 80 degrees north. There were however two key differences from our last flight to the equator and back. One, I was the only one of my three-person-team on board (we’ve always flown two of us before). Two, we were not coming back (at least not for a while). This is the first leg of the round-the-world part of the journey. The plan was simple: Anchorage, Alaska, via as-far-north-as-we-can-get-with-the-fuel-we-have.
Flying up the west coast of the U.S. and into Canada was stunning. We passed above beautiful mountain ranges and volcanoes over Oregon, Washington and into Canada. We stayed at high altitude to conserve fuel for the northern part of the leg.

Mount Rainer viewed from the DC-8 flying from Palmdale, California to Anchorage, Alaska (Credit: Christina Williamson).
Mid-way above Canada we found ourselves above a heavy blanket of cloud. We could see when we passed through the jet stream, because the winds there are very strong. This jet-stream of fast moving air around the arctic acts to isolate (to a degree) the air masses north of it from those at mid-latitudes. This is part of what makes it so interesting to study arctic air. We’re interested in how much pollution from lower latitudes affects things here, and to see what happens in the atmosphere where it’s much cleaner than most places we get to sample. People have observed lots of new particle formation (where small aerosol particles form directly in the atmosphere, changing from the gas phase into a solid or liquid phase) in the artic in summer time before, and I was interested to see if we would also witness this process. I need to analyze the data before I can really say what we sampled on this flight.
Once through the jet stream we found a hole in the clouds and managed to descend. We were in the arctic, flying over broken sea ice. It was beautiful and serene. Just ice and water below and white clouds above. We saw lots of melt pools of bright blue water on the ice.

Flying over the arctic on the DC-8 (Credit: Christina Williamson).

Flying over the arctic on the DC-8. The dark and bright blue patches are melt pools in the ice (Credit: Christina Williamson).
Somewhat reluctantly we found another hole in the clouds and ascended, to stick to our brief of scanning the atmosphere, and to conserve fuel. It’s interesting to sample at all altitudes, but heading so far north and being low over the ice was very exciting for all on board. The farthest north we got was 79 degrees N, definitely a record for me.
We then headed west towards Alaska, making a missed approach over Dead Horse. The land here was interesting, lots of pools of water, and strange hexagonal patterns in the grass the I’m told are caused by permafrost melt.

Views over Dead Horse, Alaska. (Credit: Christina Williamson)
We then came farther south over Alaska, making another missed approach at Fairbanks, and eventually landing in Anchorage, where the mountains, ocean, and cool, damp air were there to greet us as a refreshing change from the Californian desert.

Me and the DC-8 having arrived in Anchorage. (Credit: Max Dollner, University of Vienna).
We had a hard down day in Anchorage. Most of this was spent servicing the instruments and processing data from the flight, but I also found some time in the evening to take a short stroll to a beautiful beach and have dinner with colleagues looking over a lake on which water-planes were frequently taking off and landing. It’s definitely a nice bonus that we get to see beautiful places like this while working.

Views during an evening stroll in Anchorage, Alaska (Credit: Christina Williamson).

Some of the ATom science team having dinner in Anchorage, Alaska. From left to right: Eric Apel (UCAR), Kirk Ullmann (UCAR), Jeff Peischl (CIRES/NOAA), Joseph Kaitch (CIRES/NOAA), Christina Williamson (CIRES/NOAA), Maximilian Dollner (University of Vienna).
Christina Williamson blogs regularly about the ATom Mission and other adventures in atmospheric science at christinajwilliamson.wordpress.com and tweets as @chasingcloudsCW.
I have been looking forward to the second leg of the ATom mission since I first worked up the plan, collaborating with the others on the leadership team and with the flight operations crew from NASA’s Armstrong Flight Center. It would start in the hot, high desert in Palmdale, California, and journey to the high Arctic, dipping low over the floating sea ice near the North Pole, before ending in Anchorage, Alaska. What an opportunity to observe the extremes of Earth’s weather and climate, atmospheric composition, and ecosystems!
We flew this mission on August 1. The contrast between the environments could not have been more dramatic. We started our work at 4 a.m. so that the DC-8 could take off before the searing heat of the day — forecast to be 110 F (43 C). A few hours later we were in one of the coldest, most remote parts of the Northern Hemisphere. It was stunning and beautiful. But also sobering, because both places are experiencing huge impacts from the warming climate and from air pollution carried there on the winds.
In the High Desert: Fires, Urban Pollution, and a Solar Farm

Solar farm in the desert north of Palmdale, CA. Credit: Steve Wofsy
The desert near Palmdale was particularly forbidding when ATom started up. Large fires in the nearby mountains blanketed the region with choking smoke as ash sifted down from the sky. Increasingly frequent fires are an expected consequence of climate warming in this area. Even after the fires died down, the atmosphere over the desert was hazy with urban pollution originating in the Los Angeles Basin. It was unnerving to be in the midst of these events.
But as we departed the desert, I could see huge solar and wind farms generating carbon-free electricity. This sight was much more hopeful. If we apply our minds and resources to the problem, we can make significant progress in slowing the increase in atmospheric CO2 and generate new economic value at the same time. It is a long process though—a generational challenge.
In the High Arctic: Melting of Floating Ice, and More Fire Influence on the Atmosphere
We flew for several hours in the DC-8 and finally reached our destination at 80o N, the top of the world. The weather was very unfavorable at first, with heavy cloud cover obscuring the surface and making it unsafe for the DC-8 to fly low. I was really worried that we would miss the chance to sample the atmosphere and see the fabled ice cap. Then we found breaks in the clouds, and we could see the broken ice floes at the surface. We flew down to 500 feet (150 m) for a close encounter.
These images are deeply disturbing. Here we are, almost at the North Pole, and the floating ice is broken up, with dark water between flat slabs that are covered with dark covered melt ponds. Dark features accelerate melting of the floating ice by absorbing sunlight and soaking up the warmth that radiates from the atmosphere (“longwave radiation”). As melting increases the areas of open water (called “leads”) and melt ponds, more energy is absorbed and melting is accelerated. We can see with our own eyes the phenomenon known as “positive feedback”: warming and melting feed on each other, accelerating the disappearance of the ice cap.

Our first glimpse through the clouds of the floating ice at 79 N on 1 August 2016. Credit: Steve Wofsy

Close-up view of ice floes at 79 N from 500 feet. The dark features on the ice are melt ponds, and the dark areas of between the floes are open water of the Arctic Ocean. The floes are very flat, signifying “first year ice”. Credit: Steve Wofsy
The flatness of the ice is also shocking. It is “first year ice” that formed last winter from the freezing of open water. We never saw any of the lumpy “multi-year ice”, which used to survive for many years when the climate in the high Arctic was colder than it is today. A lot of ice that we saw at 80o N yesterday will be gone by the end of the melting season in September. I don’t know if we will have an open ocean so close to the pole this year, but the ice that we saw certainly didn’t look very resilient.
The coverage of floating ice has shrunk dramatically in the Arctic ocean over the past 35 years (that’s just ½ of my age!), as shown in the chart below. In the 1980s, the whole Arctic ocean was ice-covered at the end of winter, and about half remained ice-covered at the end of the melt season. In the 2000s, this picture abruptly changed. There was no longer full coverage at the end of winter, and, by the record low year of 2012, late summer ice cover had been reduced to only one half of its former extent. That is a drastic change to the global environment, in just the time since my children were born.
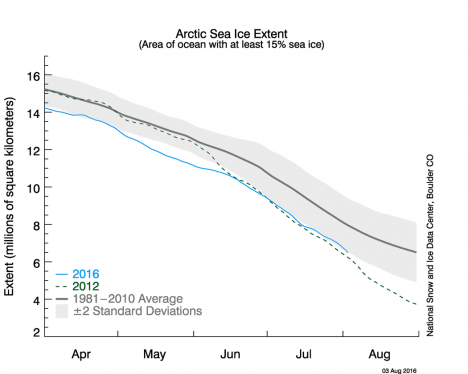
Chart of Arctic Sea Ice Extent showing the summer melt season for 1981-2010 average (black), record low in 2012 (dashed green), and this year 2016 (blue). Credit: National Snow and Ice Data Center
In 2016, as you can see from the chart, we are on track to match that record low ice extent from 2012. The high Arctic really is the “point of the spear” when it comes to climate change. But very few of us get to see the ongoing changes with our own eyes. The scientists and crew flying on the DC-8 are really privileged to have witnessed this phenomenon first hand, even though it was unsettling to observe it.
The atmosphere at 80o N had very low levels of pollutants. But once we turned around and headed for the Alaskan coast, our instruments reported strong signals from the huge fires burning the boreal forest in Siberia. This pollution was pervasive for hundreds of miles before we made landfall at Deadhorse, Alaska—the start of the Trans-Alaska oil pipeline. Viewing the huge oil and gas facilities in Deadhorse certainly gave a contrasting impression compared with the solar farms in California!
Volcanoes on Our Route – Mother Nature is Really Amazing

Mount Rainier, Washington. Credit: Steve Wofsy
I will close on a more positive note. On the way from California to the Arctic, we flew by the stunning volcanoes of the Northwest U.S. These iconic forms remind us of the beauty and overwhelming power of Mother Nature. Our Earth is really awe inspiring, and worth protecting.
Photo Credits: Steven C. Wofsy, on board the NASA DC-8 in the ATom project, August 1, 2016.

Steve Wofsy (ATom PI), Bruce Daube and Róisín Commane with the DC-8 in a nice and cool hanger in Palmdale, CA.
I’m Róisín Commane, a researcher at Harvard University working with Steve Wofsy (ATom PI) and Bruce Daube (chief engineer) to measure CO2, CH4, N2O and CO concentrations during ATom. As the project evolves, I’m hoping to provide some insight into both the science we are doing and what it means to work on an aircraft mission as unusual as ATom. Moving to a new place every few days will be tiring but I do love an adventure! I will be updating @ATom_Harvard on Twitter regularly with interesting insights, photos and videos if you want to follow along in real time.
We have been based in Palmdale, California as we prepare for the start of ATom. Palmdale is technically in Los Angeles county but is located just north of the San Gabriel mountains, about 60 miles north of Los Angeles. It’s at an altitude of 800 m (~2000 ft) and is a high desert zone so summers are brutally hot and dry and winter temperatures hover around freezing.
I find there is something enchanting about the rich morning light of the desert. But then the heat arrives and I have to retreat to places much cooler. The heat in Palmdale is tough for me to deal with. While the desert dryness should be easier to deal with than the humidity of Boston (my US home town) on a bad day, temperatures up to 40oC (110F) are just brutal for anyone. So I hide inside in the middle of the day and wear SPF 60 when I venture out for brief periods. The hanger is so large that it stays a lot cooler than outside but it does mean that we have had to start pretty early for all our flights here or many of our instruments will overheat before take off (as happened on Test flight #1). Thankfully, the temperatures cool rapidly as we fly at high altitudes and on the equator flight, I was absolutely frozen!
The schedule for our flight to Anchorage, Alaska, is slightly eye-watering for me as I’m somewhat of a night owl normally. We will be collected at our hotel at 4:00 am with all our bags packed by the wonderful Jim Elkins from NOAA who offered to do a second run to the hanger just for us. I already dropped back the rental car so we’re a little short on transport options. The aircraft will be opened up to the science crew at 4:30 am so we can start to prepare our instruments. At 6:00 am we will have a crew briefing and at 7am the aircraft doors will close. Then at 7:30am we will take off for northern (and hopefully colder!) lands.

The DC-8 on the ramp at NASA Armstrong for our sunrise boarding. Credit: Christina Williamson
Last Friday, July 29, was the first real science flight of the mission. Driving down to the base at 4am, Mumford and Sons’ ‘Cave’ blasting through the stereo, I definitely felt excited (and a little sleepy). On-board we powered up the instruments and ran through our pre-flight checklist. We’d made some changes the previous day to allow an instrument whose inlet had to be taken off the plane because of vibrations, to sample through ours. When we ran air through a filter into our system to check for leaks, we saw a small one had appeared. Thus ensued a tense 20 minutes of checking through and tightening the new fittings, but in the end we managed to get rid of the leak.

Pre-flight meeting in the lab at NASA-Armstrong before the flight with crew, pilots and science team. Credit: Christina Williamson
After a quick meeting in the hanger, where we discussed the flight plan and went over science goals and safety procedures, we were good to go. This mission is different from most aircraft-sampling campaigns because we are not chasing particular features, like biomass burning plumes or storms, but just aiming to get representative slices of the ‘normal’ atmosphere, whatever that may be.

Agnieszka Kupc, NOAA/University of Vienna running our suite of instruments measuring aerosol microphysical properties and Dan Murphy, NOAA, running an instrument looking at the chemical composition of individual aerosol particles during the ATom equator flight. Credit: Christina Williamson
Taking off the air was hazy and dusty, but soon got cleaner as we headed out over the Pacific. The flight plan was a simple one: get over the ocean, fly south to the equator, and then come back again, all the while going up and down as many times as fuel allows. In the end that made for 8 profiles. There’s not much change of scenery on a path like this, just sea and clouds, but the unchanging, rather featureless expanse has a calming kind of beauty to it.

A typical view from the equator flight on the DC-8. Credit: Christina Williamson
Although there is not a lot in terms of visual features, the data coming in from my instrument shows a host of variation hidden at sizes invisible to the human eye. There are times when all of the particles are really small, and we can infer (although we need to carefully check this from the data later) that these have formed recently from the gas phase in a process called nucleation. Sometime we pass through a layer of much larger particles on an ascent or descent. These can be pollution layers transported out from the continents, or they could be salt from the ocean or transported (again, combining our data with lots of data from other instruments is necessary before we can say anything for sure).

View at the bottom of a low profile near the equator. Credit: Christina Williamson

Haloed shadow of the DC-8 on the clouds below. Credit: Christina Williamson
Another exciting thing about being on a research plane is that we get to see more of how the plane is flown than on commercial flights. When it’s safe we can get permission to go up and sit in the extra seat in the cockpit. I got to sit in it for landing, which was really exciting. The perspective from there is very different from back in the body of the plane. The pilots are very busy, communicating with the different air-traffic-control stations as they come in to land, and guiding the plane in. The runway seems to come up exceedingly fast and looks impossibly small until we’re right on it.

In the cockpit during landing back in California. Credit: Christina Williamson
From the equator, we’ll be heading up north over the arctic on Monday, but for now, after 10 hours of flying, we’ll be taking a couple of days back at base in California to ready the instruments for the rest of the trip and analyze the data we’ve got so far.
Christina Williamson blogs regularly about the ATom Mission and other adventures in atmospheric science at christinajwilliamson.wordpress.com and tweets as @chasingcloudsCW.
In the last two weeks, we’ve wrapped up the test flights and major preparations for NASA’s ATom mission. ATom is an airborne science experiment aboard the DC-8 flying laboratory that will study the most remote parts of the Earth’s atmosphere. We want to learn how much pollution reaches areas most people would consider untouched by human influence, and to understand how pollutant chemicals in these distant regions affect things we care about.
I’m Steve Wofsy, an atmospheric scientist at Harvard University and ATom’s Principal Investigator. I couldn’t be more excited to get underway. ATom is the realization of an idea to sample cross-sections of the world’s atmosphere that I have dreamed about for more than 20 years. The idea for ATom started in the 1990’s, when I participated in a series of aircraft missions that characterized the chemistry of the Earth’s stratosphere, spanning from the Arctic to the Antarctic. Our images of global chemical changes were immensely powerful in helping us to understand depletion of stratospheric ozone. Together with my colleagues in those stratospheric missions, Paul Newman of NASA’s Goddard Space Flight Center and Michael Prather of University of California, we started to think about how we might undertake such a study in the troposphere, where we all live.

The Los Angeles skyline is hazy with smog generated from air pollutants emitted by cars. The air pollution doesn’t stay in the city, however, but will eventually travel to other parts of the world. Credit: Michael Prather
The chemistry of the atmosphere is very important for human beings and for all creatures on Earth. In the ATom study we want to understand air pollution and climate change. Air pollution has immediate and long-term consequences for human health, agriculture, and ecosystems. Climate change in the era of fossil fuel emissions and industry is being driven mostly by the gases we pump into the atmosphere.
To understand the chemistry of the atmosphere, we have to be able to measure many different chemicals in tiny amounts. The only way to do that is by going out in the atmosphere itself. We can’t make the measurements only at the ground, because the atmosphere extends far above the surface. Some measurements can be made using satellites that look down into the atmosphere, but many measurements must be made directly in the air, because the concentrations of many important gases are far too small, and the patterns we need to observe are too fine, to be measured from space.
Scientists have been using airplanes to sample the atmosphere since the 1940’s, when Britain’s Royal Air Force flew missions to understand the occurrence of contrails, which are simply water vapor that condenses behind an aircraft in certain atmospheric conditions. Today’s modern flying laboratories take sophisticated instrumentation throughout the atmosphere, in locations remote and nearby, polluted and nearly unpolluted. It is a very exacting, challenging endeavor, but one that pays off for both our understanding of the planet and for protecting it and the people on the ground.
Some of the most daring measurements of atmospheric chemistry were made in the stratospheric missions I mentioned before, between 1985 and 2000. We used the high-flying ER-2 aircraft carrying extremely sensitive instruments to measure very reactive “free radical” chemical species – in particular the chemicals responsible for the hole in stratospheric ozone that was forming each year over Antarctica. The measurements cannot be done by capturing air and taking it back to the lab – these chemicals are constantly generated by sunlight, and then they react and vanish in seconds, minutes, or hours.

NASA’s ER-2 aircraft takes scientific measurements from 65,000 feet. It was instrumental in understanding the depletion of stratospheric ozone. Credit: NASA Armstrong Flight Research Center
The insights scientists were able to gain because of those data sets led to huge societal changes, including the Montreal Protocol, the international treaty banning a whole class of industrial chemicals that were harming Earth. The amazing global consensus to ban these chemicals could not have happened without the ER-2 aircraft measurements.
Those data did not come easily. Scientists had to develop a whole new class of instruments that had to operate autonomously, since only the pilot is on board the ER-2. The plane had to go to the limit of its capability to sample in very inhospitable locations. Pilots, engineers and scientists worked very closely together to ensure safe execution of these flights.

ATom’s Principal Investigator Steve Wofsy in front of NASA’s DC-8. Credit: Tom Ryerson
The equipment on board the DC-8 in ATom likewise has to be very carefully engineered to be both safe and exquisitely sensitive and accurate. Because the chemistry of the troposphere is much more complex than that of the stratosphere, the number of independent instruments has multiplied to 22. The crew has to manage this very complex payload, with many possible hazards. The plane will often be flying very low to the ocean surface, and it will cross some of the most remote and inhospitable areas on the planet. Many of the instruments in ATom have their heritage in those flights of the ER-2.
The preparation of the DC-8 and the science payload has been meticulous. The planning of flights and logistics around the globe for our ten stopovers was undertaken to meet multiple constraints of safety, cost, and science objectives. We don’t know yet what we’ll see, but it will be the most detailed slice of the atmosphere we’ve ever measured. We hope to find out how human beings are changing the global atmosphere, what are the effects on climate, and what these changes may mean for the health and welfare of human beings and global ecosystems. We are confident in our readiness, eager to head out on our adventure, and ready to report back.